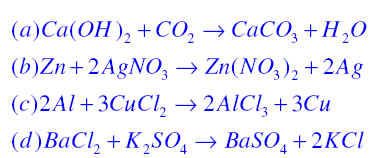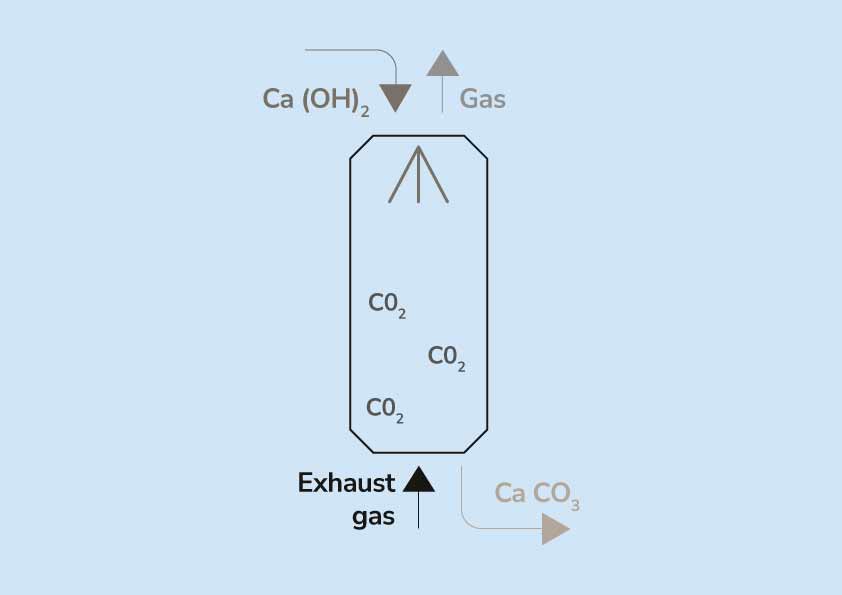
SOLVED:When carbon dioxide is bubbled through a clear calcium hydroxide solution, the solution appears milky. Write an equation for the reaction and explain how this reaction illustrates that CO2 is an acidic

science chemistry precipitation reaction carbon dioxide | Fundamental Photographs - The Art of Science
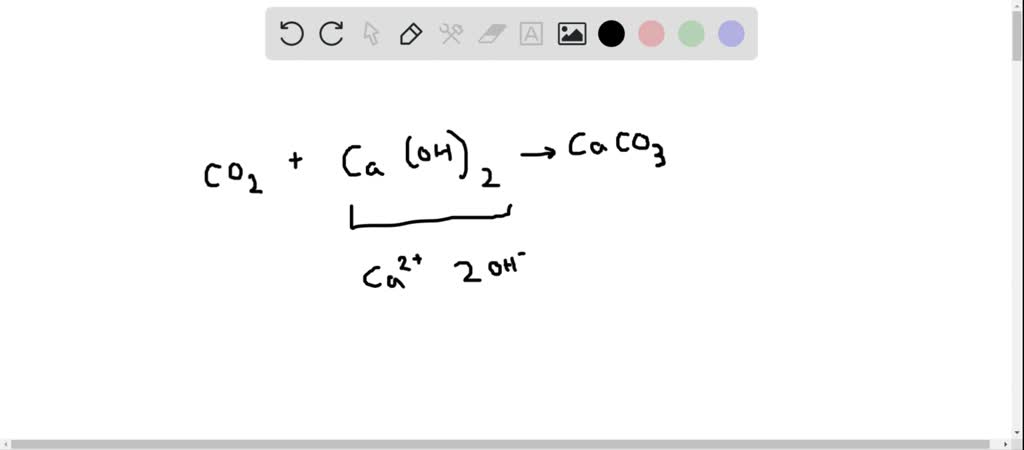
30. Ca(OH)_2 + CO_2 CaCO_3 + H_2O Why calcium hydroxide + Carbon dioxide form Calcium carbonate + Water

Hydroxide + carbon dioxide is equal to calcium carbonate + water balance the equation numerically - Science - - 12540547 | Meritnation.com

Energy-efficient chemical regeneration of AMP using calcium hydroxide for operating carbon dioxide capture process - ScienceDirect

Write the balanced chemical equations for the folowing reactions (a) Calcium hydroxide + Carbon - YouTube

EP0610781A1 - Carbon dioxide absorber and its manufacturing method using concrete sludge - Google Patents
Write the balanced chemical equations for the following reactions: i. Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water - Sarthaks eConnect | Largest Online Education Community

AQA GCSE Science & Additional Science Chemistry 1 Topic 2 Hodder Education Revision Lessons Limestone and building materials Click to continue. - ppt download

Write the balanced chemical equations for the following reaction:Calcium hydroxide + Carbon dioxide → Calcium Carbonate + Water.

Write the balanced chemical equations for the following reactions.A Calcium hydroxide + Carbon dioxide → Calcium carbonate + waterB Zinc + Silver nitrate → Zinc nitrate + SilverC Aluminium + copper chloride

Passing Carbon Dioxide Gas Through Calcium Hydroxide Solution Activity Diagram Drawing Easily - YouTube

Reactions of carbon dioxide - Gas chemistry - (CCEA) - GCSE Chemistry (Single Science) Revision - CCEA - BBC Bitesize